Nearly 2 million Americans are diagnosed with cancer every year, and approximately half of them will develop metastatic bone disease (MBD). As evolving systemic therapies improve life expectancy, the prevalence of oncology patients requiring primary or revision arthroplasty will increase, as will the need for durable reconstructions to improve their quality of life.
Independently, the frequency of primary hip and knee replacements is also growing, with current estimates predicting more than 2 million procedures annually by 2030. As such, orthopaedic surgeons will increasingly be needed to care for oncology patients with pathologic fractures, infection, and other complications.

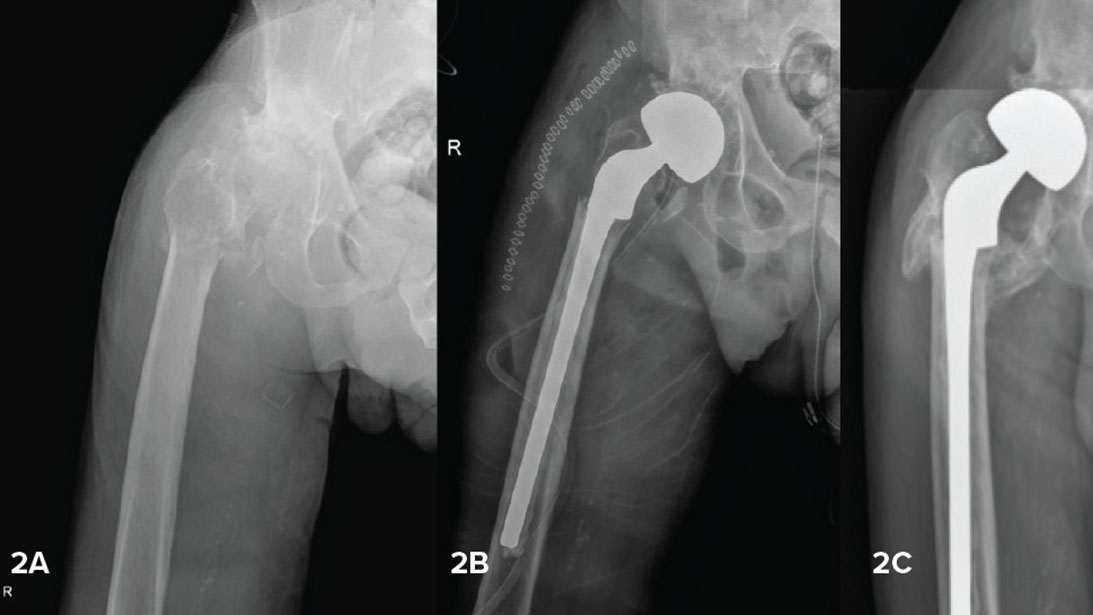
Surgical management of femoral neck fractures
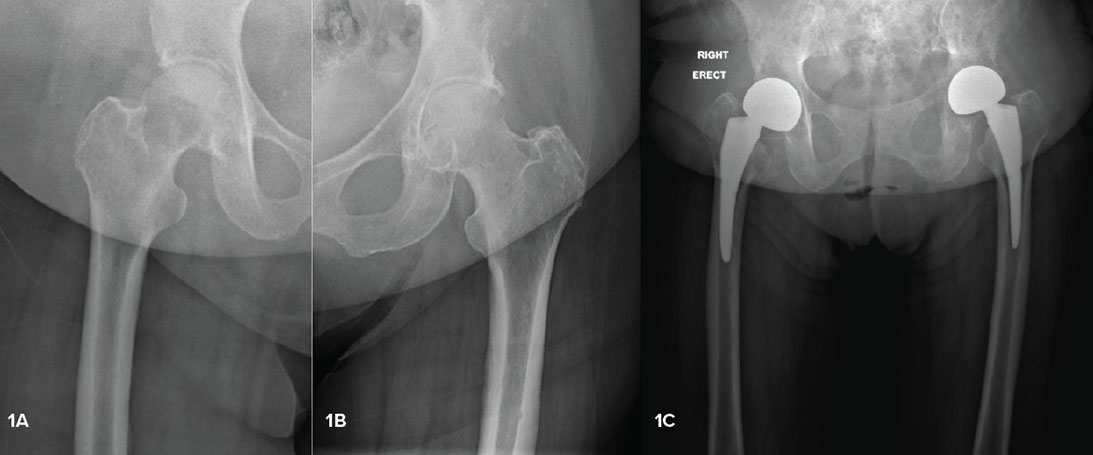
Impending and completed pathologic fractures involving the femoral neck/ head should be treated with hemi- or total hip arthroplasty. Internal fixation is not a reliable option due to poor bone quality and risk of progression, leading to a high risk of implant failure. There is no clear consensus about the use of hemiarthroplasty (HA) versus total hip arthroplasty (THA) for patients with proximal femoral metastases. HA and THA have been previously found to be equivalent in terms of reoperation, mortality, estimated blood loss, transfusion rate, length of stay, prosthetic joint infection, periprosthetic femur fracture, and patient-reported outcome measure, according to the Pathologic Fracture Group.
Acetabular wear and its implications are potential concerns in HA. Good prognosis, primary bone tumors, younger age, and longer follow-up are all associated with revision for acetabular wear; however, the risk is lower in the setting of metastatic disease, and some patients who develop acetabular erosion may be asymptomatic. The 10-year cumulative incidence for conversion to THA for acetabular wear is reported to be low, around 5% in some studies, whereas the cumulative incidence of death is reported to be much higher, around 70%. The outcomes for acetabular components are also reported to be excellent, with published survival rates of approximately 95% at two years.
As both HA and THA provide good outcomes, other factors, such as stability, should be considered. Dislocation is more common in HA performed for oncologic reasons than non-oncologic indications, so appropriate precautions should be taken intra- and postoperatively. In addition, previous reports have found higher rates of dislocation in THA compared to HA, although the instability events often occur in patients with more complex problems that require acetabular reconstruction and sometimes proximal femoral replacement (PFR).
When arthroplasty is deemed appropriate, implant options should be considered with respect to the specific situation. For example, calcar-replacing implants may be helpful for certain intertrochanteric lesions or fracture patterns. Long-stem prostheses were historically used to bridge the entire bone; however, in the absence of existing distal disease, the increased risk of complications such as cardiopulmonary events outweighs the prophylactic benefit. Similarly, while cemented fixation was once the standard of care in MBD, modern stem design has broadened the indications for cementless fixation. The Multicenter Orthopaedic Tumor Research (MORTaR) Group evaluated outcomes after HA versus THA in cemented versus uncemented stems. While there were too many differences between the two groups to make direct comparisons, the authors concluded cemented fixation is reliable and well-selected patients may be candidates for uncemented femoral stems (patients <65 years old with adequate proximal femoral bone quality and after removal of all macroscopically visible proximal disease), regardless of radiation status. In these authors’ experience and opinion, a press-fit stem is acceptable if the pathology is radio-sensitive and solid rotational stability can be achieved intraoperatively.
Considerations for inter- and subtrochanteric fractures
In contrast to femoral head/neck lesions, which should be managed with arthroplasty, inter/subtrochanteric lesions can be managed with internal fixation, calcar-replacing femoral stems, or megaprosthesis, depending on the situation. Of note, 50% or more of pathologic fractures will not heal due to poor bone healing potential related to underlying disease as well as side effects of radiation and systemic treatment. As such, some form of arthroplasty rather than intramedullary stabilization leads to more reliable outcomes, particularly in the setting of radioresistant histologies and/or large metastases with significant bone loss. Many factors should be considered in this decision; when in doubt, consultation with or referral to an orthopaedic oncologist may be most appropriate.
Renal cell carcinoma (RCC) metastases present a unique situation because this histology is often radioresistant, increasing the risk of disease progression and mechanical failure. In addition, these tumors may be highly vascular, so preoperative embolization is often used to reduce blood loss. En bloc resection of the proximal femur in RCC has been recommended due to potential survival benefits; however, the literature on this topic is inherently subject to selection bias, so the decision to resect versus stabilize RCC metastases remains debatable.
Periprosthetic fracture management
Both arthroplasty and trauma surgeons may encounter pathologic periprosthetic fractures, and the impaired biology and potential for local tumor progression must be taken into account when considering open reduction internal fixation (ORIF) of a Vancouver A or B1 fracture. A Vancouver B2 or B3 fracture is most reliably managed by revision to an implant that will be durable and functional for the patient, often a proximal femoral replacement. However, under certain circumstances, a B2 fracture could be managed with rigid fixation that will be able to withstand cyclical loading in the absence of biologic healing.
Perioperative management requires a multidisciplinary team
Metastatic cancer is complex and requires the expertise of a multidisciplinary team. With rare exceptions, all patients should undergo radiation of any bone metastases managed with intralesional surgery (i.e., stabilization or arthroplasty that does not involve wide resection of the tumor). Radiation decreases the risk of local disease progression and recurrence; therefore, it may reduce implant failure as well as tumor-associated pain. Unless patients receive radiation preoperatively, they should be scheduled for consultation with radiation oncology soon after surgery, with the goal of starting treatment once the wound is healed.
Medical oncology should also be engaged to help direct perioperative cessation and resumption of systemic treatments (chemotherapy, immunotherapy, hormone replacement therapy, etc.). Bisphosphonates should be considered as a useful adjunct in decreasing skeletal-related events in MBD, and denosumab may have even better efficacy than zoledronic acid in this population.
Patients with MBD are at increased risk for thrombotic events, so postoperative anticoagulation regimens should be more aggressive than for healthy non-oncologic arthroplasty patients (i.e., low molecular weight heparin or direct oral anticoagulant rather than low-dose aspirin).
Finally, as oncologic pain is often chronic, severe, and multifactorial, pain care specialists are an excellent resource in the treatment of postoperative and cancer-related pain.
Conclusion
Various forms of hip arthroplasty may be indicated for proximal femoral MBD, and the optimal treatment depends on several patient- and disease-related factors. Pathologic fractures are at high risk for nonunion due to reduced mechanical strength, poor biologic healing capacity, systemic therapies, and perioperative radiation, making arthroplasty solutions more reliable than ORIF in many situations. Recognizing the considerations that are unique to pathologic fracture management will help orthopaedic surgeons offer cancer patients durable solutions to restore mobility and maximize quality of life.
Lily Wood, MD, is an arthroplasty fellow at Oregon Health Sciences University. Cara Cipriano, MD, MSc, FAAOS, is an associate professor and chief of orthopaedic oncology at the University of Pennsylvania.
Rina Jain, MD, FAAOS, is an attending surgeon at Synergy Orthopedic Specialists and health sciences clinical instructor at the University of California, San Diego.
References
- Group USCSW. U.S. Cancer Statistics Data Visualizations Tool. Released June 2025.
- Swanson KC, Pritchard DJ, Sim FH. Surgical treatment of metastatic disease of the femur. J Am Acad Orthop Surg. 2000;8(1):56-65. doi:10.5435/00124635-200001000-00006
- Lesensky J, Blecha O, Vcelak J, Belzarena AC. Proximal femoral replacement or revision stem hemiarthroplasty for metastatic disease of the proximal femur — is there any notable difference? J Am Acad Orthop Surg Glob Res Rev. 2025;9(8). doi:10.5435/JAAOSGlobal-D-24-00243
- Shichman I, Roof M, Askew N, et al. Projections and epidemiology of primary hip and knee arthroplasty in Medicare patients to 2040–2060. JBJS Open Access. 2023;8(1). doi:10.2106/jbjs.Oa.22.00112
- Axelrod D, Gazendam AM, Ghert M. The surgical management of proximal femoral metastases: a narrative review. Curr Oncol. 2021;28(5):3748-3757. doi:10.3390/curroncol28050320
- Weber K, Randall R, Grossman S, Parvizi J. Management of lower-extremity bone metastasis. J Bone Joint Surg Am. 2006;88(Suppl 4):11-19. doi:10.2106/JBJS.F.00635
- Alvi HM, Damron TA. Prophylactic stabilization for bone metastases, myeloma, or lymphoma: do we need to protect the entire bone? Clin Orthop Relat Res. 2013;471(3):706-714. doi:10.1007/s11999-012-2656-1
- Bulut H, Sullivan MH, Rose PS. Comparative evaluation of treatment strategies for femoral metastases: endoprosthesis reconstruction versus intramedullary nailing. Eur J Orthop Surg Traumatol. 2025;35(1):273. doi:10.1007/s00590-025-04395-1
- Ratasvuori M, Sillanpaa N, Wedin R, Trovik C, Hansen BH, Laitinen M. Surgery of non-spinal skeletal metastases in renal cell carcinoma: no effect of preoperative embolization? Acta Orthop. 2016;87(2):183-188. doi:10.3109/17453674.2015.1127726
- Wodajo F, Colman M, Getty P. AAOS clinical practice guideline summary: treatment of metastatic carcinoma and myeloma of the femur. J Am Acad Orthop Surg. 2023;31(3):e118-e129. doi:10.5435/jaaos-d-21-00888
- Lawrenz J, Chenard S, Winter E, et al. What is the cumulative incidence of femoral stem revision and stem complication in cemented and uncemented hip arthroplasty for proximal femoral metastatic bone disease? Clin Orthop Relat Res. 2025;483:1652-1664.
- Reif TJ, Strotman PK, Kliethermes SA, Miller BJ, Nystrom LM. No consensus on implant choice for oligometastatic disease of the femoral head and neck. J Bone Oncol. 2018;12:14-18. doi:10.1016/j.jbo.2018.02.006
- Schneiderbauer M, Rose P, Sim F. Dislocation rate after hip hemiarthroplasty in patients with tumor-related conditions. J Bone Joint Surg Am. 2005;87(8):1832-1838.
- Hayden BL, Varady NH, Abdeen A, et al. No difference between hemiarthroplasty and total hip arthroplasty in the treatment of pathologic femoral neck fractures. J Arthroplasty. 2021;36(11):3662-3666. doi:10.1016/j.arth.2021.06.015
- Theil C, Mollenbeck B, Gosheger G, et al. Acetabular erosion after bipolar hemiarthroplasty in proximal femoral replacement for malignant bone tumors. J Arthroplasty. 2019;34(11):2692-2697. doi:10.1016/j.arth.2019.06.014
- Houdek M, Rose P, Ferguson P, et al. How often do acetabular erosions occur after bipolar hip endoprostheses in patients with malignant tumors and are erosions associated with outcomes scores? Clin Orthop Relat Res. 2019;477(4):777-784.
- Stevenson J, Kumar V, Cribb G, Cool P. Hemiarthroplasty proximal femoral endoprostheses following tumour reconstruction: is acetabular replacement necessary? Bone Joint J. 2018;100-B(1):101-108.
- Lex J, Evans S, Parry M, Jeys L, Stevenson J. Acetabular complications are the most common cause for revision surgery following proximal femoral endoprosthetic replacement: what is the best bearing option in the primary and revision setting? Bone Joint J. 2021;103-B(10):1633-1640. doi:10.1302/0301-620X.103B10.BJJ-2020-2480.R1
- Geraets SEW, Bos PK, van der Stok J. Preoperative embolization in surgical treatment of long bone metastasis: a systematic literature review. EFORT Open Rev. 2020;5(1):17-25. doi:10.1302/2058-5241.5.190013
- Gainor BJ, Buchert P. Fracture healing in metastatic bone disease. Clin Orthop Relat Res. 1983;(178):297-302.
- Yoon B, Park S, Roh Y. Open reduction and internal fixation for Vancouver B1 and B2 periprosthetic femoral fractures: a proportional meta-analysis. Hip Pelvis. 2023;35(4):217-227. doi:10.5371/hp.2023.35.4.217
- Haider T, Hanna P, Mohamadi A, et al. Revision arthroplasty versus open reduction and internal fixation of Vancouver type-B2 and B3 periprosthetic femoral fractures. JBJS Rev. 2021;9(8). doi:10.2106/JBJS.RVW.21.00008
- Stoffel K, Blauth M, Joeris A, Blumenthal A, Rometsch E. Fracture fixation versus revision arthroplasty in Vancouver type B2 and B3 periprosthetic femoral fractures: a systematic review. Arch Orthop Trauma Surg. 2020;140(10):1381-1394. doi:10.1007/s00402-020-03332-7
- Khan T, Grindlay D, Ollivere B, Scammell B, Manktelow A, Pearson R. A systematic review of Vancouver B2 and B3 periprosthetic femoral fractures. Bone Joint J. 2017;99-B(4 Suppl B):17-25. doi:10.1302/0301-620X.99B4.BJJ-2016-1311.R1
- Townsend PW, Smalley SR, Cozad SC, Rosenthal HG, Hassanein RE. Role of postoperative radiation therapy after stabilization of fractures caused by metastatic disease. Int J Radiat Oncol Biol Phys. 1995;31(1):43-49. doi:10.1016/0360-3016(94)e0310-g
- Drost L, Ganesh V, Wan BA, et al. Efficacy of postoperative radiation treatment for bone metastases in the extremities. Radiother Oncol. 2017;124(1):45-48. doi:10.1016/j.radonc.2017.05.010
- Jing D, Wang X, et al. Management of pain in patients with bone metastases. J Pain Res. 2023;13:1156618. doi:10.3389/fonc.2023.1156618