Sickle cell disease (SCD) is an inherited hemoglobinopathy characterized by a structural abnormality of hemoglobin that results in significant systemic and musculoskeletal complications. Sickle cell anemia (SCA), the most common and severe form of the disease, arises from a point mutation in the β-globin gene, leading to the production of abnormal hemoglobin S (HbS). Unlike normal adult hemoglobin (HbA), HbS polymerizes under deoxygenated conditions, forming rigid intracellular fibers that distort erythrocytes into their characteristic sickle shape. These deformed red blood cells exhibit reduced deformability, impaired microvascular flow, and a tendency to obstruct capillaries, resulting in chronic ischemia and repeated vaso-occlusive episodes.
SCD predominantly affects individuals of African ancestry and is significantly less prevalent among populations of European descent. In the Dominican Republic, where the population is largely of mixed ancestry — including African, European, and Tano heritage — it is estimated that approximately 10% of individuals carry the disease. Patients with SCD place substantial demands on healthcare systems, and optimal management requires a coordinated, multidisciplinary approach focused on the prevention and treatment of systemic complications. Care is typically led by a hematologist, with referral to orthopedic specialists when musculoskeletal manifestations, particularly progressive hip pain, develop.
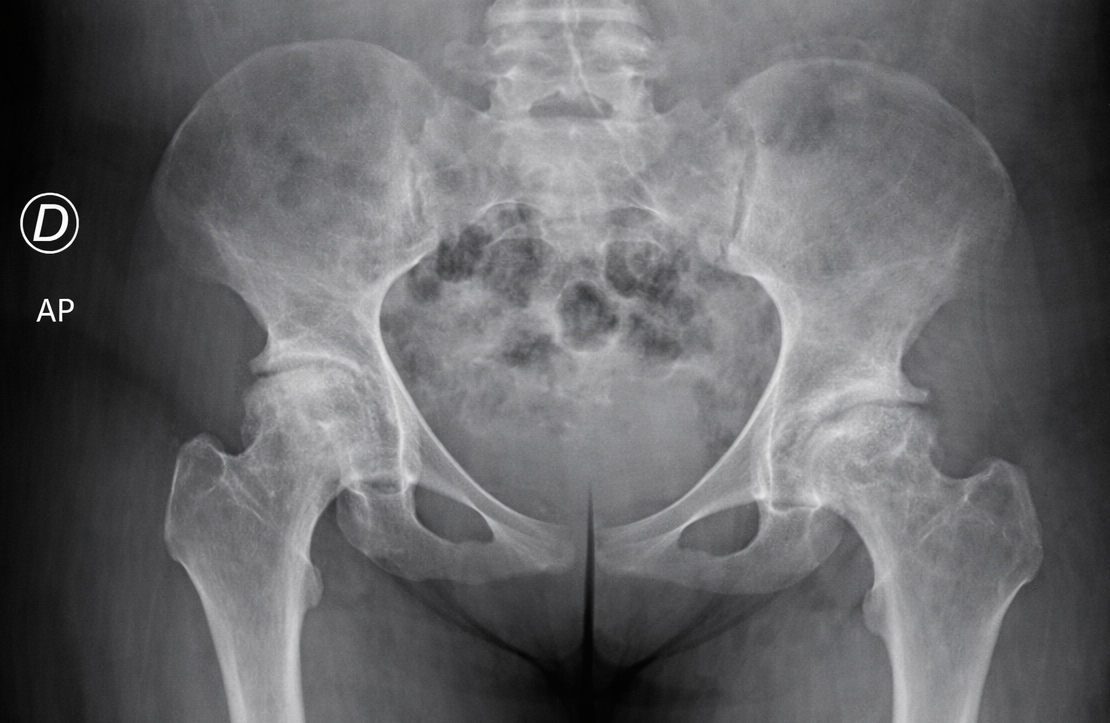
Osteonecrosis of the femoral head (Figure 1) is one of the most common and debilitating orthopedic complications of SCD. It is estimated that nearly 50% of affected patients will experience avascular necrosis of a major joint during their lifetime, and a significant proportion will ultimately require total hip arthroplasty (THA). These patients often present with a distinctive phenotype, characterized by a slender body habitus, low muscle mass, elongated limbs, and an overall asthenic appearance. From a metabolic and skeletal standpoint, chronic hemolytic anemia, marrow hyperplasia, and reduced bone mineral density are frequently encountered.
Surgical planning for THA in this population is particularly complex. Repeated episodes of bone infarction often result in a narrow, sclerotic femoral canal with dense cortical bone, making femoral preparation technically demanding. Careful attention must be paid to the femoral entry point and canal preparation to minimize the risk of intraoperative fracture. Appropriate implant selection Ñ particularly femoral stem geometry and fixation method Ñ is critical to reduce the risks of early loosening and mechanical failure. Additionally, extensive soft tissue releases may be necessary to restore adequate range of motion and achieve balanced hip mechanics following implantation.
Beyond the technical challenges of surgery, patients with SCD have an underlying prothrombotic and inflammatory state that necessitates vigilant postoperative management. Follow-up care should be multidisciplinary and more prolonged than that of the general arthroplasty population. Key objectives include prevention of vaso-occlusive crises, avoidance of acute chest syndrome, and optimization of perioperative anemia under the guidance of hematology specialists. These patients also demonstrate an increased susceptibility to periprosthetic joint infection, underscoring the need for meticulous surgical technique, careful perioperative monitoring, and close long-term surveillance Ñ particularly in cases of staged or simultaneous bilateral hip arthroplasty.
Postoperative rehabilitation is an essential component of care and should be structured and closely supervised. Physical therapy not only facilitates functional recovery of the replaced joint but also plays a critical role in minimizing pain crises involving other joints, a risk that may be heightened in patients with preserved splenic function.
Although total hip arthroplasty in patients with sickle cell disease presents significant perioperative and technical challenges, it remains a necessary and often life-changing intervention. The early-onset disability associated with hip osteonecrosis can profoundly limit education, employment, and quality of life, forcing many patients to relinquish much of their youth and productivity. When clear indications are present, joint replacement should therefore be pursued thoughtfully and meticulously, with an appreciation of both the biological complexity of the disease and the profound functional benefits that successful intervention can provide.
Delfilio Martinez, MD, is a pediatric orthopedic surgeon, specializing in hip and knee arthroplasty at TraumaCare RD; the chief of service at the Dr. Hugo Mendoza Pediatric Hospital; and the arthroplasty fellowship director at Ney Arias Lora Hospital.
Jose A. Taveras, MD, is an orthopaedic surgeon specializing in hip and knee replacement surgery at Clinica Corominas in Santiago, Dominican Republic. He is a past member of the International Committee of the AAOS.
Elido Perez, MD, specializes in hip and knee joint replacement. Rehabilitek/Hospital Daro Contreras. He is also a board member of the Dominican Society of Orthopedics and Traumatology (SDOT).